Oncomine Dx - A Companion Diagnostic (CDx) solution for identifying target gene mutations in cancer.
Indication for Testing
- Detecting acquired genetic variants arising in tumors associated with solid cancers.
- Detecting up to 2,769 variants across 50 clinically significant cancer genes, including: Single Nucleotide Variants (SNV) in 45 genes, Copy Number Variations (CNV) in 14 genes, and Gene Fusions in 19 genes.

Intended Patients
1. The test is indicated for patients with solid tumors, where identifying genetic mutations is essential for targeted therapy selection and personalized medicine.
- Lung Cancer: ALK, BRAF, EGFR, ERBB2, KRAS, MET, NTRK1, NTRK2, NTRK3, RET, ROS1.
- Breast Cancer: AKT1, BRAF, ERBB2, ESR1, NTRK1, NTRK2, NTRK3, PIK3CA, PTEN, RET.
- Gastric Cancer: BRAF, ERBB2, NTRK1, NTRK2, NTRK3, RET.
- Cholangiocarcinoma (Bile Duct Cancer): BRAF, ERBB2, FGFR2, IDH1, KRAS, NTRK1, NTRK2, NTRK3, RET.
- Colorectal Cancer: BRAF, ERBB2, KRAS, NRAS, NTRK1, NTRK2, NTRK3, RET.
- Melanoma: BRAF, KIT, NRAS, NTRK1, NTRK2, NTRK3, RET, ROS1.
- Thyroid Cancer: BRAF, NTRK1, NTRK2, NTRK3, RET.
- Ovarian Cancer: BRAF, ERBB2, NTRK1, NTRK2, NTRK3, RET.
- Hepatocellular Carcinoma (Liver Cancer): BRAF, NTRK1, NTRK2, NTRK3, RET.
- Pancreatic Cancer: BRAF, ERBB2, KRAS, NTRK1, NTRK2, NTRK3, RET.
- Over 70 other solid tumor types.

2. Patients at stages requiring targeted therapy guidance / personalized medicine
-
Patients about to initiate treatment.
-
Patients with non-responsive or resistant to standard treatment.
-
Patients with relapse / progression requiring re-assessment of genetic profiles.
3. Patients with suitable biopsy samples
-
Requires limited Formalin-Fixed Paraffin-Embedded (FFPE) tissue samples with 10–20% tumor content – from only 10ng of DNA/RNA – and as few as 2 FFPE sections.
-
Patients unable to provide tissue biopsies who require liquid biopsy sampling.
4. Clinical contexts requiring legally valid results and treatment guidance
-
Referencing clinical practice guidelines (ESMO, EMA, FDA, NCCN).
-
Selection of targeted therapies.
-
Supporting formal clinical decisions with the legal validity of CDx testing.
Technical Specifications
- Specimen Types: FFPE tissue (10% tumor content, 10ng DNA/RNA), liquid biopsy (20ng cf-TNA).
- Technology: Targeted Next-Generation Sequencing (NGS).
- Gene Panel:
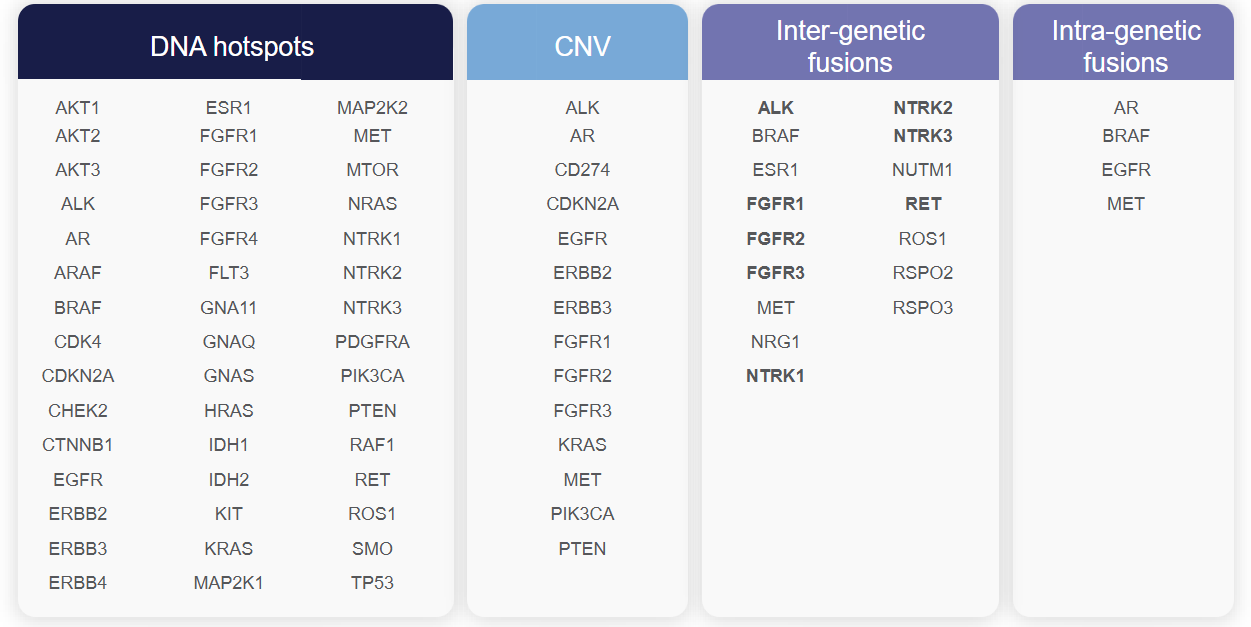
- Number of Genes: 50
- Number of Variants: 2,769
- Variant Types: SNV/Indel, CNV, Fusion
- Sensitivity: 95–99%
- Specificity: 99%
- Turnaround Time (TAT): 3–5 days









